Describe bohr's model of the atom3/7/2024 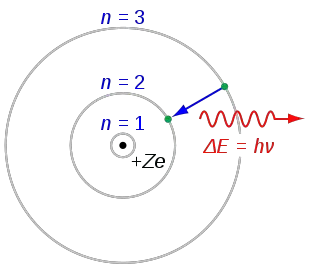
(Image credit: Pixabay) The Bohr model, introduced by Danish physicist Niels Bohr in. The model was proposed by physicist Niels Bohr in 1913. A model of an atom according to Niels Bohr. The Bohr Model is a structural model of an atom. Unfortunately, there was a serious flaw in the planetary model. The Bohr model is neat, but imperfect, depiction of atom structure. This does not satisfy the Heisenberg uncertainty principle, which is that it is not possible to know the position and momentum of a particle simultaneously.\): Niels Bohr with Albert Einstein at Paul Ehrenfest's home in Leiden (December 1925). Rutherford experiment with alpha particles shot at a thin gold foil resulted in the. Its value is obtained by setting n 1 in Equation 6.5.6: a0 40 2 mee2 5.29 × 10 11m 0.529 Å. The radius of the first Bohr orbit is called the Bohr radius of hydrogen, denoted as a0. Thomson actually) which blew a big hole in 'Plum Pudding Hypothesis' of the atom. The electron’s speed is largest in the first Bohr orbit, for n 1, which is the orbit closest to the nucleus. Thomson that resulted 'Plum Pudding' model of the atom and the 2nd one by Rutherford (a student of J.J. The main problem lies in the idea of electrons in circular orbits. Well there were two experiments back to back one by J.J. In principle the quantisation aspect of the model is still believed to be correct. So it was not possible for electrons to occupy just any energy level. Bohr model is utilized for giving a lucid explanation to the features of an atom. 
The electrons were in orbits around the nucleus, held in their orbits by the electric force that attracts negatively charged electrons to the positively charged nucleus. Niels Bohr proposed a new atomic model by giving a better explanation to the Rutherford model. Soon, people proposed a planetary model of the atom. He believed that electrons moved around the nucleus in circular orbits with quantised potential and kinetic energies. This small massive positively charged object is called the nucleus. The word ‘atom’ actually comes from Ancient Greek and roughly translates as. 9.4: The Bohr Model - Atoms with Orbits is shared under a CK-12 license and was authored, remixed, and/or curated by Marisa Alviar-Agnew & Henry Agnew. It also fails to explain zeeman effect when magnetic field splits a spectral line. In fact, we have to go all the way back to Ancient Greece to find its genesis. It fails to prove the Heisenbery uncertainity principle. Though our graphic starts in the 1800s, the idea of atoms was around long before. (b) The energy of the orbit becomes increasingly less negative with increasing n. 2: The Bohr Model of the Hydrogen Atom (a) The distance of the orbit from the nucleus increases with increasing n. In Rutherford's model most of the atom's mass is concentrated into the centre (what we now call the nucleus) and electrons surround the positive mass in something like a cloud.īohr's most significant contribution was the quantisation of the model. This graphic takes a look at the key models proposed for the atom, and how they changed over time. In this state the radius of the orbit is also infinite. Siyavulas open Physical Sciences Grade 10 textbook, chapter 4 on The atom covering 4.2 Models of the atom. Bohr’s model of the hydrogen atom started from the planetary model, but he added one assumption regarding the electrons. Schrodinger, Heisenberg, Born and many others have had a role in developing quantum theory. 
Bohr thought that electrons orbited the nucleus in quantised orbits.īohr built upon Rutherford's model of the atom. One of the most important contributions to atomic theory (the field of science that looks at atoms) was the development of quantum theory. Rutherford described the atom as consisting of a tiny positive mass surrounded by a cloud of negative electrons.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
